It just won’t stop cropping up. After I returned from giving the talk in Manchester, checking through the news in the world of science, not only did I come across impressive progress in coastal research (my number 6 unknown ), but there, in Wired Science, was a report titled “Baffling Patterns Form in Scientific Sandbox.” It began “With nothing more than beads in a glass box, physicists have revealed yet another mysterious property of granular solids, now recognized by scientists as a unique state of matter, like solids or gases” - my number 2 in the ten things we don’t know about sand: “What is it?”
The report describes work by Ralf Stannarius and Frank Rietz of the Otto von Guericke-University Magdeburg in Germany; using, as is so often the case when investigating the bizarre behaviours of granular materials, very simple apparatus, they have produced some quite astonishing results. Results that demonstrate, yet again, that we don’t really know how to describe granular materials - they are not consistently solids or liquids, but rather a state of matter in their own right. We don’t really know how many states of matter there are - in addition to the solids, liquids, and gases, there are numerous other candidates, including liquid crystal, amorphous, Bose-Einstein condensates, and, of course, the notorious quark-gluon plasmas (all this, I will readily admit, from Wikipedia, where I also discovered something called a “superglass” - but the article was categorised as an “orphan”). As Heinrich Jaeger, the granular guru, remarked when talking about his recent work on the nano-scale forces causing droplet formation in falling streams of sand, these “experimental results open up new territory for which there is currently no theoretical framework.”
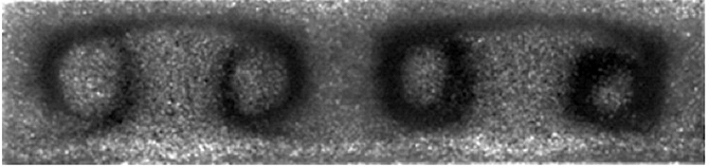
The fact that granular materials do not like to remain mixed has long been observed, and Stannarius and Rietz describe earlier experimental work which their latest research continues. Put a granular mixture in a container and vibrate, shake, rotate, or otherwise disturb it and it will perpetrate an act of self-segregation. Stannarius and Rietz have shown that such a mixture not only self-segregates, but convects. In the image at the top of this post (from their publication), it almost seems as if the material is mocking us, saying “hey, you’ll need better spectacles than these through which to understand what we’re up to” (OK, blogger’s poetic license). “On the Brink of Jamming: Granular Convection in Densely Filled Containers” by Rietz and Stannarius was first published last year, but last month they released an update with compelling and extraordinary videos of granular convection. The apparatus was, indeed, simple, but clever. As shown below, they used a simple mix of two sizes of glass sand-sized beads in a container that is essentially a Hele-Shaw cell, just like the home-made one I have used in my kitchen physics experiments. Once filled (and it is important that the cell is filled beyond a critical level), it’s placed in a mechanism that simply rotates it, slowly, for hours. The cell is back-lit and photographed periodically.
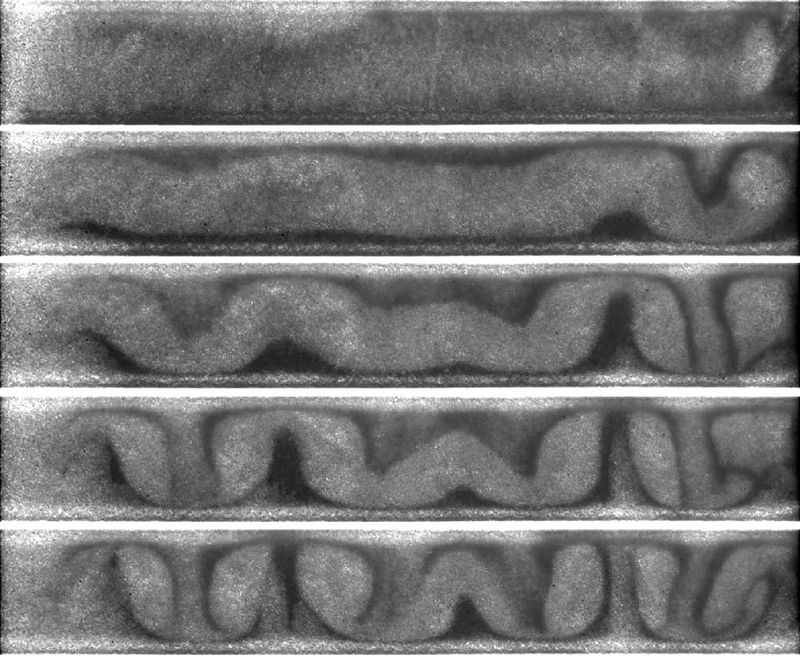
The results are staggering. The movies, of various runs of the experiment and at various resolutions, can be found at the Magdeburg website (caution: if you go to the MIT Technology Review report, the movie link embedded there is for a 150 mb version - better to check out the link I’ve just mentioned). The movies are dramatic and well-worth watching, but just to whet your appetite, I’ve assembled a time-series of stills, shown below.
In a completely unpredictable (and unpredicted) way, the grains develop a graceful series of dynamically sustained convection rolls. As the authors wrote, “Known mechanisms for granular convection could not be applied.” Another echo of Heinrich Jaeger’s comment that “Physicists have a rich toolbox for dealing with solids, liquids and gases. But we don’t have a manual for when the old categories don’t apply.” Whatever next in the mysterious world of granular materials?

Comments
Fascinating. Imagine what might happen in these very homogeneous mixes if you varied shape and particle density. Or would the effect disappear?
Yes indeed - the range of grain size does make a difference, but convection is still seen in an essentially “monodisperse” granulate (albeit reduced in amplitude): “For that purpose, a cell was filled with the nearly monodisperse beads of the large species (<10% radius variation). It is evident … that a convection structure of similar wavelength as in the mixture is formed, but with considerably lower flow amplitude.” (from the original paper). As for shape and density, there’s obviously a lot of fun work to be done - we do know that something similar happens with poppyseeds and natural sand, but more controlled experiments are needed.