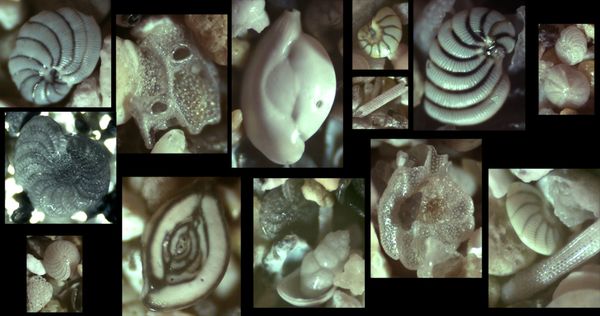
When I wrote a while ago about the sands of Kefalonia and the mysteries of where exactly the homeland of Odysseus lay, I commented that one of the samples was clearly crammed with critters. However, at the time, I did not have the wherewithal to examine them in any great detail, this requiring more sophisticated scrutiny than my hand-held digital microscope, however clever, could offer. But now I am united with the wherewithal: a full-blown digital microscope on which I had splurged shortly before leaving the UK, and which has now completed its sedate journey to southeast Asia. I’ve been spending some escapist time playing around with it, and it occurred to me that the critters of Kefalonia would make ideal subjects for a series of portraits. In addition, a number of readers of this blog have asked about sand grain microphotography, and so, amateurish though my efforts are, I thought that this would be a good opportunity for at least a few thoughts.
So today’s post is really on two topics: the wonders of the biogenic components of sand and how to experience those wonders. The sand of  .
.
But there are other critters in this Kefalonian sand, and, since the depth of my microfaunal expertise stretches no further than my knowledge of, say, string theory, perhaps I should open this up for more knowledgeable readers to engage in a “name that critter” session.
So now to the means of granular portrait photography. My new toy (like the hand-held low power digital microscope I described earlier) is made by Celestron; this blog does not do commercials, but I have to say that this is an impressive piece of kit.
It’s the “LCD Professional Digital Microscope Model # 44345.” It is, like most microscopes within the reach of the amateur, primarily for biological  . Images are recorded in a 1Gb internal storage and can be downloaded by a USB connection to your computer; there is also an SD card slot.
. Images are recorded in a 1Gb internal storage and can be downloaded by a USB connection to your computer; there is also an SD card slot.
The images are excellent, but it’s critical to have image manipulation software (I use Adobe Photoshop) for processing and refinement. I find that the colour balance of the original image has a green emphasis; this can easily be adjusted in Photoshop, as can light levels, cropping, and so on. With my previous system – a standard binocular optical microscope with a digital camera attachment – a custom white balance could be set up, but I don’t think that there’s a way of doing this with the digital microscope.
There is, of course, the inherent problem of a limited depth of field with 3D objects such as sand grains. The microscope has an excellent fine focus system, and so the optimal image can be carefully selected, but there are always parts of the image that are not in focus. I have recently been investigating what would seem to be some clever software, available online, that deals with this problem by selecting the in-focus parts of each of a successive series of images of an object and re-combining them into a single image; if anyone has experience of this, your views would be much appreciated (it would seem to work for landscape photos as well as microscopic images).
So, there we are, my latest toy. It’s a great instrument and, although, perversely, I would like one smaller magnification, it’s providing much pleasure, lots of surprises, and some decent images. Any comments welcome!
Meanwhile, some transmitted light portraits:
[For the ultimate high-tech microphotography of sand grains, see Gary Greenberg’s site and book; but many arenophiles are also much more skilled than I am – for example, Catalin Stefan’s World Atlas of Sands website has great photographic galleries]


Comments
Focal stacking software is good for dealing with the depth of focus issue; my Nikon software that goes with my Nikon digital scope can take a series of photos from different focal depths and turn them into a single crisp image. It’s a nifty trick for three-dimensional samples.
Nice forams! I use the Helicon Focus software which does a good job of focus stacking images of rock cuttings, which are more or less the same scale as your sand samples. I find that stacks of between 4 and 6 images are usually sufficient for cuttings at 10x or 12x (binocular microscope magnification).
Callan and Howard - thanks for the comments and the encouragement! Helicon is the software I have been looking at, so I think that, once I can find the time, I will go ahead and give it a try.
Ooh, critters! I’ve never seen backlit photos of forams before, they’re even more mysterious and beautiful.
In the mid-late '80s I spent some time on the white beaches of Florida (gulf region), didn’t know anything about tiny shelly creatures, and undoubtedly tramped over thousands of these things. Ah, to go back and do over – I’d be looking at that sand under a microscope, for sure!
beautiful pictures… a microscope and grains…i always feel a surge of excitement!
“And every chambered cell,
Where its dim dreaming life was wont to dwell,
As the frail tenant shaped his growing shell,
Before thee lies revealed,–
Its irised ceiling rent, its sunless crypt unsealed!”
Beautiful, just beautiful. Thank you.
Hi Michael,
thanks for your interesting post on foraminifera in the sand.
Imaging of forams is not an easy task as tininess and thickness result in a quite limited depth of field. Furthermore internal structures shining through and surface features cannot be illustrated in just one 2-dimensional image.
You show the big guys unfortunately many forams are smaller and in the fraction of 100-400µm. In a full sample from the ocean bottom (not beach) tens or hundreds of such small ones come with a big one.
For my foraminifera.eu project we use:
SEM (see a Planorbulina image - you illustrated one - at http://www.foraminifera.eu/planorbulina-mediterranensis2-socotra.html) SEM only shows the surface.
Optical Imaging with stacking using Helicon Focus (works for us only sufficiently down to 400µm) (see e.g. a 30Mya old http://www.foraminifera.eu/nodosaria-vertebralis6-kobrow )
Drawings: if not altered from the original, we view it as state of the art (see e.g. http://www.foraminifera.eu/brady.php
Slicing and than imaging the flat surface: http://www.foraminifera.eu/nodosaria-intermittens4-kobrow.html (slicing of single specimens is time consuming, so we don’t do it a lot)
3D modelling - not yet done and probably out of reach for us amateurs.
So our view is to use different imaging techniques for the different purposes, with realistic drawings as the best solution.
BTW beyond the technical issues of photographing is the whole story of foraminifera such as its recording in the sediment year per year the conditions of the world oceans. Thus forams are widely used in climate reconstruction, paleoenvironmental studies and oil/gas exploration.
Cheers
Michael Hesemann
Foraminifera.eu Project
Hamburg, Germany